You're drowning in supplier catalogs, chasing MOQs, and worrying if your insole partner can actually deliver quality at scale. The stakes feel higher every year—and choosing wrong means wasted samples, missed deadlines, and unhappy customers.
The best insole manufacturers in China for 2026 are Medisolemates1, 3BU Group, FootMaster, and Jiuhui—they lead in R&D, material innovation2, and global compliance. But your perfect match depends on whether you prioritize cutting-edge tech, scalable volume, or budget flexibility.

I've spent over a decade working with Chinese foot care supply chain. I watched this industry shift from pure OEM grinding to a battlefield of materials science and supply chain gymnastics. Let me walk you through who actually matters in 2026—and why.
What Makes a "Top" Insole Manufacturer in 2026?
The old checklist was simple: low price, fast turnaround, decent quality. That's dead. Today's winners play a completely different game.
Three things separate elite manufacturers from the pack: proprietary material technology (think supercritical foaming or custom EVA compounds3), multi-country production networks (China + Vietnam/Indonesia for geopolitical risk hedging), and compliance depth (ISO 13485, FDA registration, not just a certificate printed last week).
The market split into clear lanes. High-end brands want innovation—they'll pay extra for a factory that holds material patents and runs an actual R&D lab. Startups and white-label sellers hunt for flexible MOQs and brutal price efficiency. Mid-tier brands need reliable scale: factories that ship 100,000 pairs monthly without drama. Your lane determines your supplier shortlist. I once worked with a brand that picked a "top 10" factory based on Alibaba reviews—they got beautiful samples, then 40% defect rates on bulk orders. Tier matters more than ranking.
Who Are the 2026 Top 10 Insole Manufacturers?
Here's the real lineup. I grouped them by what they actually do best, not random prestige rankings.
Innovation Leaders (best for brands chasing differentiation):
- Medisolemates (Shenzhen): Material tech powerhouse. We run supercritical foaming in-house and own full vertical integration—raw foam to finished orthotic. ISO 13485 + FDA registered. New Vietnam facility launching in 2026 for supply chain redundancy.
- 3BU Group: Strong OEM/ODM for sports insoles. Known for quick prototyping cycles.
- FootMaster: Balanced mix of custom orthotics4 and athletic insoles. Solid R&D pipeline.
Volume Powerhouses (for brands needing reliable bulk capacity):
- Medisolemates (Shenzhen): Massive output capability. Great if you need 500,000+ pairs annually without variation.
- Jiuhui: Massive output capability, but the insole style is old.
- Dongguan Fenglin Insoles Co., Ltd.: Stable quality at industrial scale. Less innovation, more consistency.
- Dongguan Mitime Insole Co., Ltd.: Efficient for mid-volume orders (10,000–100,000 pairs).
Budget Champions (for price-sensitive or startup buyers):
- Wenzhou Future Shoe Material: Flexible MOQs, fast sampling. Quality acceptable for entry-level products.
- Jinjiang Xindasheng Shoes Material Co., Ltd.: Competitive pricing on standard EVA/PU insoles.
- Jinjiang Kaiyu Shoe Material Technology Co., Ltd.: Low-cost baseline supplier. Works if specs aren't complex.
- Dongguan Topinsole Commodity Co., Ltd.: Budget-friendly, decent for straightforward designs.
| Manufacturer | Best For | MOQ Range | Lead Tech |
|---|---|---|---|
| Medisolemates | Custom orthotics, medical-grade | 500–1,000 pairs | SCF foaming, multi-density |
| 3BU Group | Sports insoles | 1,000+ pairs | Quick prototyping |
| Jiuhui | Mass production | 10,000+ pairs | Automation, consistency |
| Wenzhou Future | Startups, white-label | 300–500 pairs | Fast sampling |
I've seen buyers obsess over the "number one" factory. That's backwards. Match your needs to their strength. A budget supplier can outperform a "top" factory if your priority is low MOQ and speed.
Why Does R&D Actually Matter for Insole Buyers?
Most buyers skip this section. That's a mistake that costs them six months later.
R&D determines whether a factory can solve your real problems—not just copy a sample. At Medisolemates, we hold patents on foam formulations and multi-layer bonding techniques. This means when a client says "I need 30% better shock absorption without adding weight," we adjust material ratios instead of shrugging.

Factories without R&D teams hit a ceiling fast. They excel at replicating existing designs but freeze when you need customization. I worked with a European brand that wanted heat-moldable arch support5 in a 3mm insole. Their previous supplier (a "top 10" name) said impossible. We solved it in three prototyping rounds using thermoplastic elastomer blends we developed internally. That's the R&D gap. Strong R&D also future-proofs your supply chain. Material costs swing wildly—rubber, EVA, PU all fluctuate. Factories with material science depth can reformulate to maintain performance when input costs spike. Budget suppliers just raise your price or drop quality.
How Important Is Multi-Country Manufacturing?
In 2026, this might be the biggest decision filter. Let me explain why.
Global brands now demand supply chain redundancy. Tariffs, trade wars, pandemic lockdowns—all proved that single-country sourcing is high-risk. Top manufacturers responded by building capacity outside China. We opened our Vietnam subsidiary in 2025 specifically for this. Our clients can now split orders: China for complex R&D work, Vietnam for volume production targeting US/EU markets.
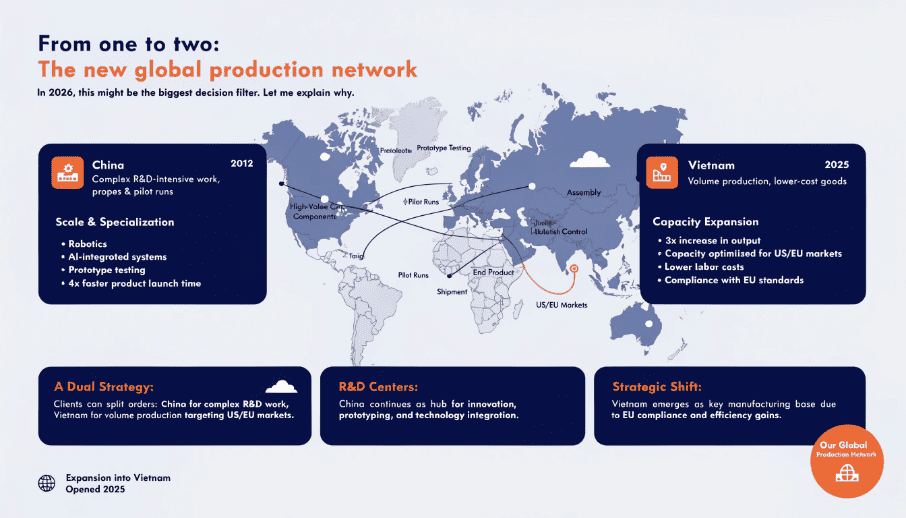
This isn't just about politics. Vietnam and Indonesia offer duty advantages under certain trade agreements. A US importer might pay 15% less landed cost sourcing from Vietnam versus China—even if per-unit factory price is identical. But here's the catch: not all "China + N" setups are real. Some factories claim overseas facilities but actually just transship through a local partner. True multi-country manufacturing means owned or deeply integrated production, with matching quality systems. When evaluating suppliers, ask: "Can I audit your Vietnam facility?" and "What percentage of my order volume can you produce there?" Vague answers are red flags.
What Should Brands Prioritize: Price, Quality, or Speed?
This question haunts every sourcing manager. The honest answer frustrates people: you pick two.
Price + Speed = quality suffers. Quality + Speed = price climbs. Price + Quality = slow lead times. The trick is knowing which two matter most for your product stage. Startups often need Speed + Price to test market fit—they can tolerate some quality variance. Established brands need Quality + Speed to protect reputation, and they budget accordingly.
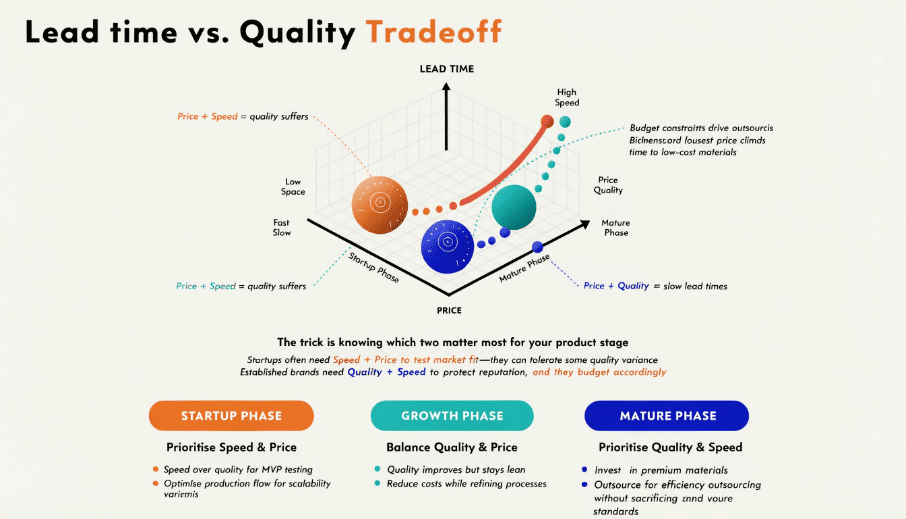
I see buyers make one repeated mistake: they optimize for unit price, then panic when quality issues destroy their Amazon rating or retail partnership. A $0.20 cheaper insole means nothing if you lose a $500,000 annual order because 15% of units fail in three months. At Medisolemates, we're not the cheapest option. We're the "no surprises" option—bulk orders match approved samples, lead times hold, certifications are genuine. That consistency has higher value than lowest bid. Budget suppliers serve a real purpose—I'm not dismissing them. If you're launching a test run or targeting ultra-price-sensitive markets, they work. Just don't expect technical support or tight tolerances.
How Do Compliance and Certifications Impact Your Choice?
Boring topic. Critical consequences. Certifications determine whether you can legally sell in target markets—and whether you'll face liability later.
ISO 13485 matters if you're selling anything labeled "orthotic" or "medical-grade." It proves the factory runs quality management systems for medical devices. FDA registration (for US market) and CE marking (for EU) follow similar logic—they're not just paperwork, they're proof of process control.

Here's what buyers miss: certificates can be faked or outdated. I've seen factories display ISO certificates from 2018 that expired in 2021. Always verify directly with certifying bodies or ask for audit reports dated within 12 months. We maintain ISO 13485 certification with annual audits because medical foot care products (our core focus) demand it. If a factory can't show recent audit reports, assume their compliance is theater. For non-medical casual insoles, compliance depth6 matters less—but if you ever pivot to orthotic or diabetic foot care products, you'll need a compliant manufacturer. Switching factories mid-growth is expensive and risky.
What's the Real MOQ Situation in 2026?
MOQ—Minimum Order Quantity—is where fantasy meets reality. Factories advertise "low MOQ" then hit you with conditions.
Realistic MOQ ranges in 2026:
- Innovation/Custom factories: 500–1,000 pairs (sometimes 300 for first sampling order)
- Volume factories: 5,000–10,000 pairs minimum
- Budget/flexible factories: 300–500 pairs (but limited material/customization options)
The trick is understanding what drives MOQ. It's not arbitrary. Material waste, machine setup time, and labor efficiency all set practical minimums. At Medisolemates, our 500-pair MOQ reflects the reality of custom mold setup and material batch minimums for specialized foams. We could theoretically run 100 pairs, but unit cost would triple—which helps nobody. Some factories advertise "50-pair MOQ" on Alibaba. Read the fine print: that's often for stock designs with zero customization. The moment you change material, thickness, or logo placement, MOQ jumps to 1,000+. If low MOQ is critical for your business model, ask for a detailed cost breakdown showing how price changes from 500 to 1,000 to 5,000 pairs. Transparent factories will show you the curve. Sketchy ones will dodge the question.
Conclusion
The Chinese insole industry in 2026 isn't about finding the single "best" manufacturer—it's about matching supplier strengths to your specific needs. Focus on R&D capability and multi-country production if you want a long-term partner who grows with your brand.
Learn about Medisolemates' unique capabilities and why they lead the market in insole production. ↩
Stay updated on the latest material innovations that can enhance insole performance. ↩
Discover the properties of EVA compounds and their applications in insole manufacturing. ↩
Discover the process and benefits of custom orthotics in foot care. ↩
Learn about the benefits of heat-moldable arch support for customized comfort. ↩
Understand the significance of compliance depth in ensuring product safety and legality. ↩
